Check out these upcoming publications utilizing LifeCanvas products and technologies! Our tissue processing methods and devices are utilized around the world to further medical and biological research, facilitating new discoveries and improving lives. The articles below are posted prior to peer-review in open-access medRxiv and bioRxiv online repositories.
Lung transplantation for pulmonary fibrosis secondary to severe COVID-19
Full text, medRxiv. Corresponding author Ankit Bharat, abharat@nm.org.
Research on the coronavirus and its health implications is key to understanding and managing the COVID-19 epidemic with early findings on treatments invaluable for saving and improving thousands of lives. This article from the Northwestern Memorial Hospital at Northwestern University reports key results on the first two lung transplants for Acute Respiratory Distress Syndrome (ARDS) related to COVID-19 in the US.
The authors used methods including histology, RNAscope (Bio-Techne), SHIELD-based tissue clearing (LifeCanvas Technologies), and RNA sequencing to investigate and characterize lung tissue from healthy donors, from diseased explants from patients receiving a lung transplant, and from patients who had died from COVID-19. Diseased lungs showed severe fibrosis similar to that of patients who had undergone lung transplantation to treat pulmonary fibrosis unrelated to COVID-19. The fibrosis likely had several contributing causes (e.g. the coronavirus, inflammation, mechanical/barometric damage from a respirator, bacterial infection) and was deemed irreversible. The authors conclude that in certain cases, lung transplantation is the best option for recovery from severe ARDS after COVID-19.
Cellular anatomy of the mouse primary motor cortex
Full text, bioRxiv. Corresponding author Hong-Wei Dong, HongWeiD@mednet.ucla.edu.
This groundbreaking article represents a step forward in the collaborative effort of the BRAIN Initiative Cell Census Network (BICCN) to map the entire mouse brain with cell-level detail by combining anatomical, functional and molecular data to create open-source, 3D brain cell atlases.
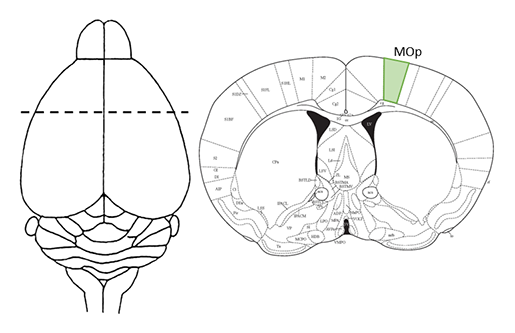
The authors use established and emerging technologies to generate a comprehensive description of the murine primary motor cortex upper limb area (MOp-ul) and identify cell types, map their distribution within the MOp-ul, and track inputs to and projections from the MOp-ul across the entire brain. Cells were labeled virally, genetically, or histologically and imaged in both 2D and 3D via a variety of high resolution techniques including the use of LifeCanvas’ SmartSPIM light sheet microscope. Computational analyses involved atlas registration, cell detection, axon density quantification, and neuronal morphology reconstruction. The datasets generated through this project are open-source and available for the benefit of researchers around the world through the BICCN. Comprehensive anatomical and functional descriptions of brain regions, and eventually a whole-brain cell atlas, will be an invaluable resource for current and future generations of neuroscientists.
Evidence for genetically distinct direct and indirect spinocerebellar pathways mediating proprioception
Full text, bioRxiv. Corresponding author Helen C. Lai, Helen.Lai@utsouthwestern.edu.
The Lai Lab at UT Southwestern researches the anatomy and development of spinocerebellar pathways related to proprioception which governs one’s sense of self-movement and where one’s body is in space. Using both traditional sectioning and 3D tissue processing methods, the group meticulously traced projections from spinal cord neurons to their terminals in the cerebellum and brainstem, thereby describing a circuit map of neurons involved in proprioception. To this end mouse hindbrain and spinal cord were processed using LifeCanvas Technologies pipeline employing SHIELD preservation, tissue clearing, and image acquisition with the SmartSPIM light sheet microscope in order to track neurons that were fluorescently-tagged within spinocerebellar circuits. Such processing and imaging of intact tissues allows for accurate tracing of axons in spinocerebellar pathways with quantification that is unbiased by region selection. Future work will focus on the organization, connectivity, and function of spinocerebellar neurons to determine how proprioceptive information is processed in the brain and spinal cord.


