What is a microtome?
A microtome is an instrument used to repeatedly slice tissue into thin, uniform sections for downstream laboratory processing and imaging. Different types of microtome are used to section different types of tissue, depending on the sample’s size, embedding medium, and preparation method (e.g. frozen or fresh).
Why is tissue sectioning important?
Depending on the application, scientists and technicians generally need to stain or label tissue for different biomarkers and then image it under a microscope to obtain useful data for diagnostic or research purposes. Sectioning can facilitate various parts of this tissue processing to data collection pipeline.
First, some tissue processing methods, such as traditional immunolabeling procedures, only work on thin sections of biological material. The probes used may not be able to penetrate thicker tissue in order to effectively bind to their targets.
Second, some imaging methods, such as wide-field fluorescence microscopy and confocal microscopy, have very limited imaging depth and thus can only acquire up to 200 µm of tissue at a time (depending on the tissue). Sectioning samples is required in order to generate higher-quality images from thicker volumes.

Lastly, some samples – such as human and nonhuman primate organs – are so large that they cannot be properly processed or imaged intact even with more advanced technologies.
How do you choose a microtome for your application?
The most commonly-used microtomes are rotary, cryostat, and vibrating microtomes.
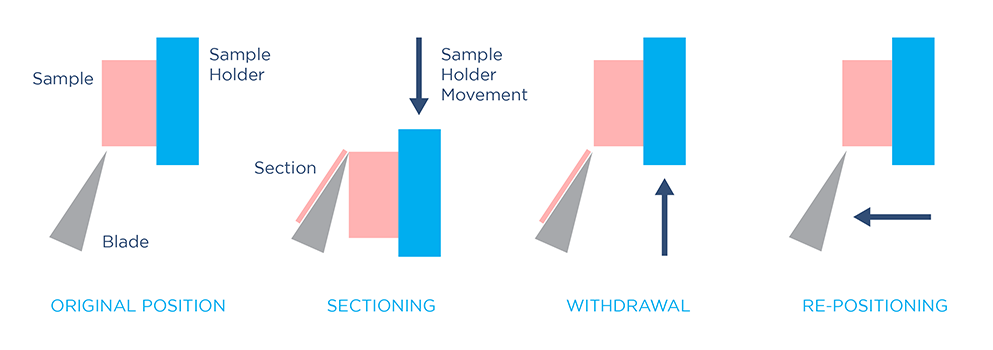
To generate sections with a rotary microtome, a sample is fed towards a fixed knife by rotating a flywheel, which can be operated manually or automatically. After each section is made, the sample holder advances by a small step to generate the next section. Rotary microtomes can generate extremely thin tissue sections (0.5 – 60 µm). However, to use this method, tissue must be hardened prior to sectioning by dehydrating and embedding the sample in paraffin or epoxy resin – a time-consuming, laborious process.
Cryostat microtomes operate similarly to rotary microtomes, except sectioning takes place in a controlled, enclosed freezer. To prepare tissue for cryosectioning, fixed or fresh samples must be embedded in a mold filled with aqueous-based medium, and rapidly frozen. Freezing and embedding the sample achieves a similar aim to epoxy or paraffin embedding: hardening the sample to stabilize it before sectioning. This allows cryostat microtomes to also generate extremely thin slices.
If you are using imaging methods that are limited to thin sections, such as electron or brightfield microscopy, rotary or cryostat microtomes are good choices. However, these methods are not efficient for imaging large samples and thick tissue.
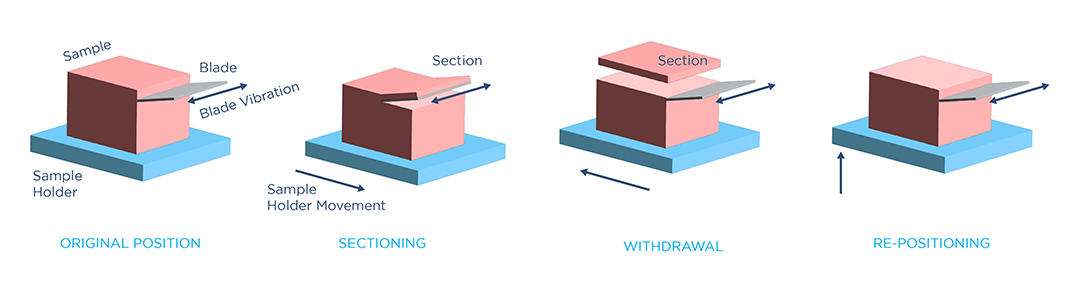
Vibrating microtomes use a vibrating blade which slowly cuts into the tissue to generate sections. These instruments can section fixed or fresh tissues directly without embedding them in a hardening medium, saving time and effort in tissue preparation. However, since they operate on soft tissue, vibrating microtomes typically generate thicker slices (>50 µm). This makes this method a more suitable tool for volumetric imaging, such as confocal, two-photon, and light sheet microscopy.
If you work with large samples like human or nonhuman primate brains, Megatome is the only instrument on the market capable of automatically, accurately sectioning tissue of this size. Similarly, if you handle large volumes of smaller samples such as biopsies, organoids, or rodent organs, only Megatome can slice large arrays of these tissues. Sectioning arrays of tissue can vastly improve your throughput while retaining slice quality and uniformity across all samples.
How does controlling blade vibration help improve the precision of tissue sectioning?
Blade vibrating frequency, amplitude, and out-of-plane blade displacement are important parameters to optimize for soft tissue sectioning. A vibrating blade exerts both shear and normal force on a soft tissue during slicing. The shear force, generated by blade vibration, performs the crucial task of “breaking” the sample at the tip of the blade by creating a critical stress and strain condition in the material. The normal force, generated by blade advancement into the sample, assists the formation of this critical condition by pushing against the tissue. However, because soft tissues can be easily deformed, this normal force must be minimized in order to prevent damage, such as tissue stretching and distortion.
"To ensure high-precision sectioning in large samples, Megatome ... minimizes out-of-plane blade displacement with an advanced vibration control mechanism."
With a high-speed shear cutting force, the critical “break” condition can be attained with less normal force and less tissue deformation – thus, increasing the blade vibration frequency leads to a cleaner cut. Megatome leverages high blade vibrating frequency and amplitude to minimize tissue damage during sectioning.
Another important factor in precise soft tissue sectioning is the out-of-plane displacement of the blade: the distance that the blade moves away from its advancing plane. This unwanted blade movement leads to tissue damage and unevenness at the slice interface, worsening as the blade length increases (and, consequently, as the sample size increases). To ensure high-precision sectioning in large samples, Megatome also minimizes out-of-plane blade displacement with an advanced vibration control mechanism.
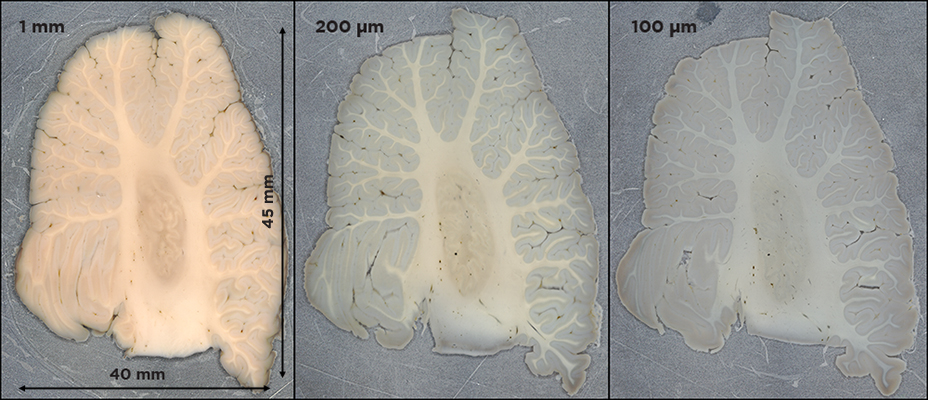
What other features distinguish Megatome from the average microtome?
The quality of tissue sectioning depends not only on blade vibration parameters, but also on the sample embedding process, and the instrument’s ease of use. Megatome comes with tools and protocols for easy and fast soft-gel embedding of tissues, including ultra-large samples and arrays, as well as automatic sectioning functions, significantly streamlining the tissue sectioning process for researchers. LifeCanvas engineers are committed to simplifying every step of the journey to high-quality 3D data – reach out today to discuss the application of our innovative technologies to your research.