Over 190 million people have now been infected with the novel coronavirus SARS-CoV-2 in the ongoing coronavirus disease (COVID-19) pandemic with a total number of deaths passing 4.1 million worldwide. Multiple organs are affected by COVID-19, however, the lungs are the main site of disease. Statistics show that 6-10% of COVID-19 patients develop acute respiratory distress syndrome (ARDS) and require urgent mechanical ventilation. Sadly, 20-40% of these acute cases lead to death.
The severity of the pandemic prompted current studies “Early outcomes after lung transplantation for severe COVID-19: a series of the first consecutive cases from four countries,” published in The Lancet Respiratory Medicine (2021) through an international collaboration between Dr. Ankit Bharat (Northwestern University, IL), Dr. Tiago N Machuca (University of Florida, FL), Dr. Mario Nosotti (Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, Italy) & Dr. Konrad Hoetzenecker (Medical University of Vienna, Austria) and “Lung transplantation for patients with severe COVID-19,” published in Science Translational Medicine (2020) by Dr. Ankit Bharat et al. (Northwestern University, IL) in collaboration with Dr. Scott Budinger (Northwestern University, IL). These publications show evidence for successful double-lung transplantation in COVID-19 patients suffering from irreversible lung injury.
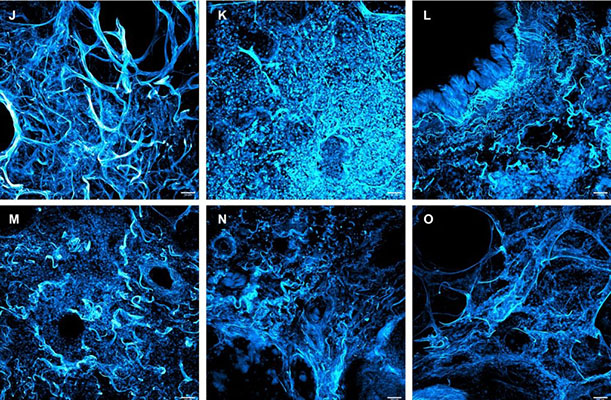
In order to evaluate the lung’s framework matrix, Bharat and colleagues obtained human lung biopsies from healthy and COVID-19 lung transplant recipients and used SHIELD tissue clearing. Sections of 100 μm thick human lung tissue underwent SHIELD preservation, followed by passive clearing with Passive Clearing Buffer. This process took 30-60 minutes for healthy lungs and ~4 hours for diseased lungs due to disorganized pattern of collagen structure and matrix in the latter.
Afterwards, cleared human lung tissues were incubated with EasyIndex to achieve refractive index homogenization and imaged. Bharat and colleagues demonstrated that explanted lung tissues from late-stage severe COVID-19 patients had extensive damage of the lung matrix framework. This was similar to other end-stage lung diseases, indicating the critical need of lung transplantation in COVID-19 patients when medical therapy is insufficient to save the patient’s life.
The integration of 3D histology was critical in these studies to enable the visualization of intricate tissue architecture. It led to a more comprehensive and accurate interpretation of pathological alterations in SARS-CoV-2 infected lungs.
Furthermore, utilizing LifeCanvas 3D tissue clearing eliminated the need for time-consuming tissue sectioning and image reconstruction. Importantly, these studies show the applicability of tissue clearing in tissues other than the brain (see recent blog post) and demonstrate how valuable 3D histology is for both human and animal studies.
Multiple studies have demonstrated successful implementation of 3D tissue clearing and imaging using LifeCanvas Technologies products and/or services. Despite the pandemic, there has been a growing trend in publications involving LifeCanvas Technologies throughout the last years. Of these publications, 30% were published in high impact journals (e.g. Cell, Nature, Science).
Finally, research groups have benefited and published through using our Contract Research services (CRO) e.g. Dr. Helen Lai’s laboratory at UT Southwestern Medical Center, TX (Pop et al., 2020 and Ogujiofor et al., 2020), as well as Dr. Nicole Calakos’ lab at Duke University, NC (Helseth et al., 2021). For this, we only require PFA-fixed samples to be sent by customers and LifeCanvas’ scientific experts complete requested end-to-end projects using SHIELD, SmartClear, EasyIndex, and SmartSPIM. This way, despite the COVID-19 quarantine and shutdown of laboratories worldwide, LifeCanvas supports research efforts to reveal critical pathways of disease progression and towards the development of treatments.
Conclusion
- Whole tissue clearing in multiple tissue types, including the lungs
- Both passive & active clearing, depending on your research goals
- Studies in both animal & human tissues
- CRO services, to continue your research and to deliver results for manuscripts and/or preliminary data for your grant submissions