Dr. Michelle Bedenbaugh started using LifeCanvas whole-brain mapping tools to explore the intersection of feeding, stress, and anxiety-like behaviors as a post-doc in Richard Simerly’s lab. In November 2024, she began her own lab at the University of Florida to delve deeper, and also examine the effects of early-life stress and aging on these behaviors.
We caught up with Michelle about her experience as a new investigator, what starting a new lab entails, and why she chose to bring LifeCanvas with her on her next big scientific journey.

You previously spoke to us as a post-doc in Richard Simerly’s lab. What is your new lab's research focus?
My lab right now has two major focuses. One of those is similar to my post-doc work, looking at the role of the melanocortin-3 receptor at the intersection of feeding and other motivated behaviors, specifically stress and anxiety-like behaviors. We also may look into how early life stress can have lifelong consequences on these behaviors, as well as related physiology and neural circuitry.
The other focus is on how responses to fasting and changes in hunger occur as animals age. We’ve started by using a whole-brain mapping pipeline to look at changes in response to fasting in young and aged mice.

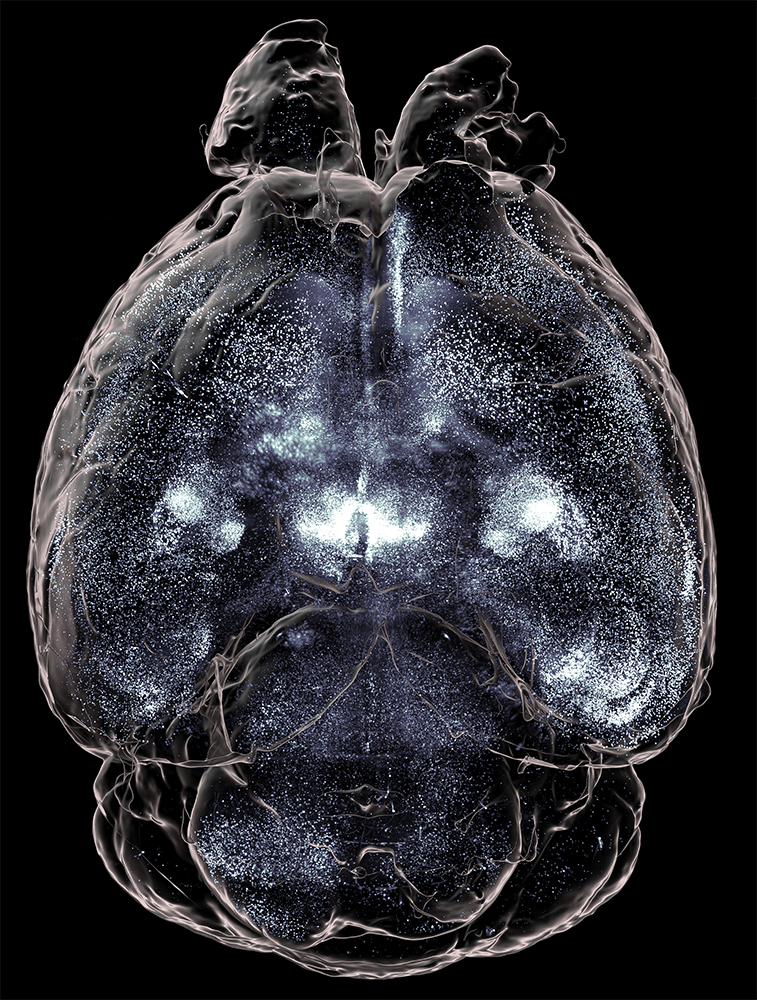
MC3R-expressing neurons throughout a whole mouse brain (right), zooming in on the hippocampus (above), imaged with SmartSPIM.
How does whole-brain mapping further your investigations?
One example is we’re using whole-brain c-FOS and microglia mapping as an unbiased approach to discovering brain areas that play a role in hunger, aging, and associated inflammation. We’ve gotten some preliminary whole brain data there already, and there are a lot of drastic changes in regions that haven’t really been implicated in hunger and energy homeostasis before. These kinds of data can allow us to find new drug targets or biomarkers for aging-related metabolic disorders.
In another example, we actually found an application that went along with an early life stress pilot we were running. The early life stress paradigm that we use is brief maternal daily separations: basically, we take pups from their dams (mothers) for four hours a day, for 10 days straight. Generally we study just the pups, but we decided to investigate the dams as well to see if there are brain-wide changes in neural activity between those who went through pup separation versus those who didn’t. We found significant changes in brain regions that deal with motivation and affect, as well as differences in oxytocin transporter expression. This is something that could be further explored and potentially used as a model for postpartum depression.
Without the whole-brain pipeline, we definitely wouldn’t have been able to make these discoveries, or even have time to consider these experiments.
Why did you choose the LifeCanvas pipeline for this approach?
I wanted to use LifeCanvas partially because I was familiar with the pipeline from my post-doc work, but also because I thought it was the best on the market for what I wanted to do. It’s very quick and easy to image one mouse brain with SmartSPIM, and the chamber is so large now that you can even line up three at a time. I’ve talked with a lot of people whose light sheet systems wouldn’t even let them image an entire mouse brain. With SmartSPIM, you can even image a whole juvenile mouse, which will become more relevant as people start to use whole-body data instead of just focusing on one organ. With the flexibility and quality of data, it was a no brainer for me.
"With SmartSPIM, you can even image a whole juvenile mouse, which will become more relevant as people start to use whole-body data instead of just focusing on one organ."
In terms of tissue processing, we really needed a SmartBatch+ to be able to do whole-brain c-FOS labeling. SmartBatch+ virus labeling also provides a high enough signal-to-noise ratio that we can use anterograde tracing to quantify synaptic terminals in whole brains.
It’s also really easy to communicate with the LifeCanvas team. When you’re setting up a new workflow, there’s always little things that go wrong in the beginning. The accessibility of the team has been very helpful in troubleshooting and getting the lab up and running.
There are so many things to consider when you start a lab. How do you prioritize and stay on track?
As you start a lab, you need to negotiate your startup funds with the university. Most of that is deciding what you need to be successful, and trying to convince them of that. No matter where you are, there’s some red tape. You have to compare different systems, and even if you know what you want to buy, you have to do some sole source justification – especially for large equipment purchases.


I prioritized getting the SmartSPIM light sheet as soon as possible, because I knew that would allow us to do a lot of preliminary experiments and ask further questions. Lots of processes, like getting an animal protocol approved, can take a lot of time to work their way through the system. With SmartSPIM, we could get started while we were waiting for everything else. We asked around the department to see if anyone had one or two brains they wanted imaged so we could train people before we had our own animals.
For equipment you’re not as familiar with, you can collaborate with other people that have that equipment and see if their system is something you’d be interested in. If it works well, their team isn’t having issues, and it’s within the right price range, you might consider it for your own lab. Even if it’s more expensive, if it’s better quality and easier to use than something cheaper, it’s worth the extra investment in my opinion. It’s going to take less time to train people, there’s going to be less user error, experiments will run more quickly … over time, that’s going to save you a significant amount of money.
How have you stayed grounded as a new investigator in a rather turbulent time for science?
The scientific community here at UF has been essential. The more senior investigators in my department are so approachable, and they’ve helped me ride the ebb and flow, and not get swept up in things that are aiming to stress us out. I’ve been trying to keep my head down and focus on asking good questions, collecting good data, and training the next generation of scientists to have the skills to do whatever they want to do. It can be very easy to get sucked into doom and gloom. I mean, it happens to me at least once a week. But being in my community and focusing on science and mentoring have helped me tremendously.


